Tomatoes are a staple crop for many farmers and gardeners alike, prized for their versatility in the kitchen and their health benefits.
Ripened tomatoes are used to make a variety of dishes and processed foods, such as pizza sauce, salsa and ketchup. To ensure the high quality of these processed, the quality of the ingredients is extremely important. However, it can be challenging to determine the perfect time to harvest tomatoes for optimal flavor and nutrition.
One of the critical factors in determining tomato maturity is pH, which can be measured using tools provided by Hanna Instruments.
Introduction to Tomato Maturity
What is Tomato Maturity?
Tomato maturity refers to the stage at which a tomato is fully developed and ready for optimal harvesting. The maturity of tomatoes is indicated by their color, size, flavor and texture.
Why is Tomato Maturity Important?
The maturity of tomatoes affects their quality, taste and shelf life. Harvesting tomatoes at the optimal maturity stage ensures they have the best flavor and texture. It also reduces the chances of spoilage during storage or transportation. Furthermore, they should be at the desired maturity before they can be used to make processed goods.
Importance of pH in Determining Tomato Maturity
How pH Affects Tomato Maturity?
While color is one way to determine ripeness, it can be inaccurate due to how subjective color is since the color of the tomato is determined by the variety of tomato. A less subjective and more accurate method to determine tomato maturity is to measure the pH of the tomato.
The pH of the tomato fruit changes during the ripening process. As the tomato matures, the pH decreases, becoming more acidic. The pH affects the texture, flavor and color of the tomato.
Benefits of pH Measurement for Tomato Growers
Measuring the pH of tomatoes can help growers determine the optimal maturity stage for harvesting. It can also help identify any problems with the soil or growing conditions that may affect the pH of the fruit.
How to use Hanna Instruments to measure Tomato pH
Preparing for Tomato pH Measurement
– To measure the pH of tomatoes, first, clean the pH meter or tester probe with distilled water.
– Rinse the tomatoes with distilled water and dry them before testing.
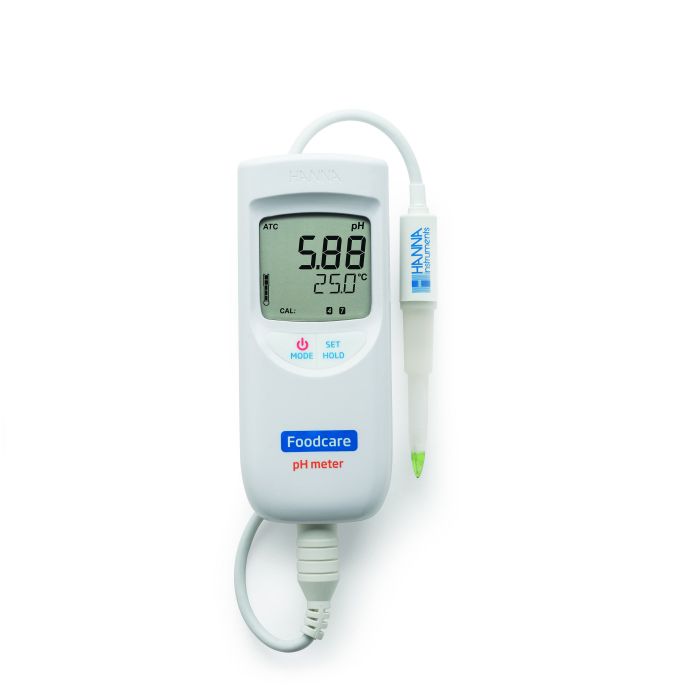
HI99161
Portable Food and Dairy pH Meter
Hanna recommends the HI99161 Portable Food and Dairy pH Meter with a FC2023 pH electrode.
The meter is small, allowing it to be easily carried, yet heavy-duty and waterproof to work in the greenhouse environment.
The FC2023 electrode is made of food grade plastic that the meets FDA regulations for direct contact with food. The conical shape of the glass bulb allows it to penetrate the tomato and the open junction provides a high area of contact with the tomato, avoiding the need to juice or homogenize the tomato for analysis.
With this shape, just by cutting the tomato with a knife and inserting the pH electrode, a pH reading can be obtained. This pH electrode is gel filled and comes with a built-in temperature sensor, both which are useful in the field.
Interpreting pH Measurements for Tomato Maturity
When it comes to determining the maturity of a tomato, pH measurement can be a useful tool. pH, or the measure of acidity or alkalinity in a solution, can give us an indication of when a tomato is ripe and ready to be harvested. But how do we interpret pH measurements and what are the target pH ranges for tomato maturity?
Understanding the pH Scale
In the case of tomatoes, a pH range between 4.5 and 5.5 is considered optimal for ripeness. As the tomato matures, its pH level decreases, becoming more acidic.
Target pH Ranges for Tomato Maturity
As mentioned, the target pH range for tomato maturity is between 4.5 and 5.5. This range can vary slightly depending on the variety of tomato and its intended use.
However, in processed food, the pH needs to be below pH 4.6 in order to avoid the growth of pathogens and microbes. Ideally, food processors aim for pH 4.2 for food safety. Tomatoes become more acidic as they mature so tomatoes are often not harvested for use in processed food until they are slightly overripe.
If the tomatoes have a pH greater than 4.2 at the time of harvest, then citric acid is added to lower the pH. The additional chemical cost of this acidification process can be reduced if tomatoes are picked at the optimal time.
Factors Affecting Tomato pH and Maturity
Several factors can affect the pH and maturity of a tomato. These can be classified into environmental and genetic factors.
Environmental Factors
Environmental factors such as temperature, humidity, soil quality and water availability can all have an impact on tomato pH and maturity.
For example, tomatoes grown in hot and dry conditions may ripen faster and have a lower pH than those grown in cooler and wetter conditions.
Genetic Factors
Genetic factors like tomato variety and seed quality can also affect pH and maturity. Certain varieties may have a naturally lower or higher pH range, while poor seed quality can result in uneven maturity and pH levels.
Conclusion and Future Applications
Determining tomato maturity by measuring pH with Hanna Instruments is a convenient and reliable method. By understanding the pH scale and target pH ranges for tomato maturity, growers can ensure they are harvesting their tomatoes at the optimal time for peak flavor and quality.
Potential Applications for Future Research
Future research could explore the relationship between tomato maturity and flavor, as well as the impact of different growing conditions on tomato pH and maturity. Additionally, studies could be conducted to develop non-destructive methods for determining tomato maturity, which would be beneficial for large-scale growers and processors.
In conclusion, measuring tomato pH with Hanna Instruments is an essential tool for tomato growers to ensure optimal yield and quality. With a better understanding of pH, growers can make informed decisions about when to harvest their tomatoes and how to maximize their overall crop.
Using the techniques and tools outlined in this article, growers can take the guesswork out of tomato maturity and achieve consistent, high-quality results year after year.
Have questions?
Contact a Hanna Technical Specialist at [email protected] or using our contact form.
AUTHOR: Tajana Mokrović, mag.nutr.