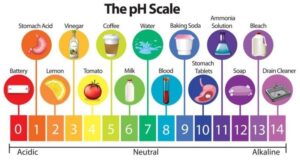
The term pH originates from a French word “pouvoir hydrogen” which means hydrogen power. It is used to describe the amount of hydrogen ions in a substance (e.g. solutions, soil). pH is the unit used to measure alkalinity of acidity of a compound, it has a scale from 0-14 pH. As the amount of hydrogen ions increase the pH of the solution decreases ( it’s becoming more acidic). At the pH 7 the solution is neutral, as the acidity increases the number decreases vice versa as the alkalinity increases the number increases. For example pH of 2 means that the solution is very acidic and pH of 10 means its very alkaline. Sorenson (1909) defined the pH and gives the pH scale: pH is the negative log of hydrogen ion (H+) activity in an aqueous solution in moles/ L. The soil with a pH of 3 has 10 times more hydrogen ions than a soil of pH 4, 1000 more pH than a soil of pH 7, 100 more than pH 6 and 10 more than pH 5.
pH influences on several key factors of soil:
- Stability on soil for crop production.
- Availability of nutrients to plants (and by that change the water availability).
- Microbial activity in the soil.
- Physical characteristics of soil, permeability, structure etc.
Different pH can alter the availability of nutrients in the soil. This is largely depended on the partition of the elements between solid and liquid phases of soil through precipitation-dissolution charges as a result of pH depended charges in mineral and organic fractions. Negative charges dominate in high pH whereas positive charges prevail in low pH. For example at the pH of 5.3, the adsorption of Cd, Cu, and Zn onto a sediment composite consisting of Al-, Fe-, and Si-oxides was 60%, 62%, and 53%, respectively. In contrast, he found that 50% of Cd and Zn adsorbed onto humic acids between pH 4.8–4.9. Cd,Cu,Al,Fe are trace elements (minimal element concentration needed for plants to function properly) so by altering the pH of soil we can improve the solubility of those elements. CO2 found in soil can alter the pH, it has a tendency to dissolve in water into H+ and HCO3- which can also lower pH and change the absorption.
The pH of soils can vary in different climates and other external factors abiotic or biotic can influence the change. Abiotic factors are: climate factors, humidity, insolation, element concentrations, type of a bedrock, exposition etc. For example in the cold and dry climate the soil is more alkaline (chernozem in steppe ) , in warm and rainy climate the soil is more acidic ( laterite in Amazon).

Soil try’s to buffer these changes in pH but sometimes some of the factors can alter this process and change the pH, it is far more common that anthropogenic factors shift the balance by using fertilizers, pesticides or similar chemical compounds. The excess of Nitrogen that came in with the fertilizer can shift the pH towards more acid state.
As we said nutrient availability differs at different pH values
Nitrogen is one of the key soil nutrients (N). Plants can absorb it in 2 different forms (NH4+ ammonium, NO3- nitrate). At around neutral pH (pH 7) microbial conversion of NH4+ to NO3- is rapid. In acid soils (pH<6) and plants with the ability to take up ammonium have an advantage.
Phosphorus availability is highly dependent on pH. When the soil is slightly alkaline or neutral the HPO4- ion is the most common as the pH lowers both the H2PO4- and HPO4- ions prevail. At higher acidities H2PO4- tends to dominate. Plants mostly absorb HPO4- ions. At pH 6-7 availability of phosphorus for plants is at the maximum.
Potassium availability tends to lower as the pH lowers because the aluminum binds with the binding spot of potassium.
Calcium and magnesium availability is more in the alkaline solutions.
Sulphate (SO42-) isn’t drastically affected by the change in pH.
Micronutrients such as manganese (Mn), iron (Fe), copper (Cu), zinc (Zn) and boron (B) are more available at lower pH.
Soil organisms and the effect of different pH values. Growth of many bacteria and actinomycetes are inhibited below pH 6. Fungi grow on a wide variety of pH, so they dominate at lower pH. Therefore the lower populations of symbionts (bacteria and some fungi) lower the nutrient availability (nitrification, n fixation). Earthworms are important for aeration and soil mixing.
The pH and other properties can shift or degrade over time, some of them by natural processes and some by human intervention that can be direct or indirect. Indirect can be by burning fuels, metallurgy, mining (can be direct), deforestation (also can be direct), waste water. Burning fuels, metallurgical processing and mining can produce a lot of green house gases, pollute with heavy metals that can all influence the occurrence of acid rain that can change the soil composition and factors. Agriculture can also change the soil especially uncontrolled use of fertilization, watering with polluted water, consistent monoculture planting etc. Fertilizers have a lot of nitrogen and phosphorus that can help the plants or even hinder their ability to grow because the more nitrogen and phosphorus (the soil is enriched) there is a bigger chance of weed (rogue) prevalence. Monocultures can deplete some nutrients and change the composition of the soil and there are two methods of overcoming this, planting different plants every year (or season) or testing and maintaining the optimal conditions of the soil.

In conclusion all of the factors are interlinked and can alter the growth and success of biota so it is very important to monitor our fields if we are growing something. As the human civilization advanced and our technological knowledge and achievements it has become easier to monitor our soils and keep them in the optimal range for specific organisms. Hanna instruments developed a lot of instruments that can help you monitor essential conditions for your plants to reach their maximum potential. There are a range of products to chose from that help you monitor the pH of your soil depending on your requirements. Starting from the testers, smaller instruments used for quick analyses of pH (there are testers varying resolutions and accuracy e.g. HI98118 Groline for solutions, HI981030 Soil Groline tester ) or even combined testers for pH/EC (electrical conductivity) / TDS (total dissolved solids). Instruments that can monitor your hydroponic nutrients 24/7 (e.g. HI981421).
HI981030- Soil pH tester
The HI981030 GroLine soil pH tester is an application specific designed pH tester for the measurement of soil pH. This meter offers many advanced features including the ability to clear any clogging of the reference junction, which results in a longer life than standard pH testers.
HI98118-Gro Line pH tester
The GroLine HI98118 pH/temperature tester is our latest pocket meter for measuring the pH of a hydroponic nutrient solution. The HI98118 has a very large easy to read LCD display that shows both pH and temperature along with calibration, stability, and low battery indicators. All operations are simplified to two buttons.
HI12922- Halo electrode for direct soil applications
The HI12922 HALO innovative state of the art electrode with Bluetooth Smart technology. It is compatible with Android and Apple devices. Easy to use wireless soil pH electrode, with the conical tip for quick and easy measurements. It has a triple ceramic junction and built-in temperature sensor. The electrode is refillable and includes a soil auger. The range of measurement is 0.00 to 12.00 pH and temperature operating range is from -5 to 70 °C.
HI389- Soil Test Kit
The chemical composition of soil includes pH and chemical elements. Soil analysis is necessary for better management of fertilization and to know the residues of fertilizers in relation to the crop, tillage and the most suitable plant choice for soil composition. An analysis can highlight shortages and help the understanding of the causes of an abnormal growth. By using the Hanna soil test, it is possible to measure pH and the most important elements for plant growth: nitrogen (N), phosphorus (P) and potassium (K).
Testing the soil during each crop cycle and comparing the results with plant growth can be a useful information for subsequent cultivations.
HI99121- Direct Soil pH meter
The HI99121 is the perfect portable pH meter for soil testing. With the HI99121 and HI12923 direct soil pre-amplified pH and temperature probe, users can test both the pH of soil directly or after preparation of a soil slurry with deionized water.
HI9814- pH/EC/TDS/T meter
HI9814 is a durable, portable pH, conductivity, total dissolved solids and temperature meter for most measurements encountered in hydroponics, aquaponics or general agriculture applications. All operations and settings, are made through only two buttons and the housing is waterproof and rated for IP67 conditions. User-selectable features include selectable TDS factors of 0.5 and 0.7 as well as auto-off after 8 minutes or 60 minutes to prolong battery life.
HI98168- pH Temperature Meter for Soil
The HI98168 is a rugged, waterproof portable pH meter that allows for the direct measuring of pH in soil. The meter is supplied with a specialize pH electrode that has rugged conical tip for soil insertion.
It has the performance and features of a benchtop meter. It has various functions: waterproof, ATC compensation, 200 h battery life, big display, PC connectivity, GLP. pH range from -2.0 to 20.0 pH and resolution from 0.1 to 0.001 pH.
HI981421-Gro Line Hydroponics monitor
The HI981421 GroLine Monitor provides 24 hour continuous monitoring of pH, conductivity (EC or TDS), and temperature in hydroponic nutrients. Quick to setup and simple to use, this monitor was designed with Hydroponics, Aquaponics, and Greenhouses in mind. Make your nutrient solution easy to manage with the GroLine Monitor and combined pH/EC/Temperature probe.
Fluctuations in your hydroponic nutrient solution can have lasting effects on your plants. The GroLine Monitor automatically logs every 15 minutes for the last 30 days, and stores min, max, and average values so you can recognize when patterns arise and help prevent future problems. For review and storage, use the USB-C to easily transfer data to a flash drive or PC using a cable. Files are exported as .cs
The versatile display of the GroLine Monitor allows for three screen modes. The LCD can display all three essential hydroponic nutrients measurements at one time, a 3-second cycle of single measurements, or a real-time graph screen with options for measurement selection and log recall.
The GroLine Monitor frees up your time by doing the testing for you. Simply set high and low alarm levels – if your hydroponic nutrient solution moves out of range a measurement error will display. A quick look at the large display will let you know if your nutrient solution needs adjusting.

HI83325-Multiparameter photometer for Nutrient Analysis
The HI83325 benchtop photometer measures 8 different key water quality parameters using 9 different methods. This photometer features an innovative optical system that uses LEDs, narrow band interference filters, focusing lens and both a silicon photodetector for absorbance measurement and a reference detector to maintain a consistent light source ensures accurate and repeatable photometric readings every time.
Consistent and thorough monitoring of plant nutrients is essential to maintaining healthy growth and reproduction. This is easy with the HI83325, a comprehensive way to monitor vital plant nutrients such as potassium, calcium and magnesium. Required in large quantities, potassium plays a vital role in water uptake and enzyme regulation. Calcium helps to strengthen plant cell walls protecting against heat stress while magnesium helps build a strong immune system.
HI83900- Suction Lysimeter
HI83900 Suction lysimeter is an ideal tool for collecting soil solution samples and then performing quantitative chemical analysis. In this way, the operator can easily monitor the level of nutrients such as ammonia, nitrate, phosphorous, potassium, sulfate, calcium and magnesium.
HI83900 Suction lysimeter is built with a porous ceramic cap connected to a transparent tube for soil solution extraction. A rubber capillary is inserted in the tube passing through a rubber cap and reaching the ceramic tip.
Lysimeter allows the extraction of a solution from the soil by creating a vacuum inside the sampler tube, that exceeds the soil water tension. This will establish a hydraulic gradient for the solution to flow through the porous ceramic cap and into the lysimeter tube. Typically, a vacuum of about -60 centibar should be drawn.
The ceramic tip of the lysimeter can be used in all types of soil. It is made of a sanitized material that does not react with the nutrients in the soil.
For better monitoring of soil solution composition throughout an entire growth period of a crop, at least two lysimeters should be installed in the root zone of a representative plant, one at the upper par and one in the lower part of the root zone.
It is recommended to replicate installations in at least two more locations, for better measurements accuracy and repeatability
HI981412-pH Dosing System
HI981412 pH Dosing System is engineered for maintaining the pH of nutrient solutions and irrigation water. The pH of nutrient solution or irrigation water is critical for the successful propagation and growth of plants. Macro and micro nutrients as well as calcium and magnesium bloom boosters require the correct pH for absorption into the plants root system. The HI981412 was developed to be an inexpensive solution for the horticulturist to maintain the ideal pH at all times. Simply insert the probe and injection valve in-line with the recirculation pump and provide the chemical to be dosed.
HI981413- Nutrient Dosing System
HI981413 Nutrient Dosing System developed to be an inexpensive solution for the horticulturist, it is engineered for maintaining the EC and TDS of nutrient solutions and irrigation water. Simply insert the probe and injection valve in-line with the recirculation pump and provide the chemical to be dosed. It is essential for plants to have stable EC and TDS of nutrient solutions to booster the growth and production of plants.